NAFDAC Warns Against Unregistered ViroActive+ Capsules Claimed to Cure HIV
The National Agency for Food and Drug Administration and Control (NAFDAC) has warned healthcare providers, patients, and the public against the use of unregistered ViroActive+ Capsules circulating in Nigeria.
NAFDAC disclosed that the report was received as a complaint at its Bauchi State office, noting that the unregistered ViroActive+ is marketed as a drug purported to cure HIV.
“It is promoted online in Nigeria as a herbal supplement claimed to boost the immune system, treat or cure viral infections, especially HIV, and improve general health.
“It is important to note that there is no credible scientific evidence that herbal capsules can cure HIV. HIV is treated with antiretroviral therapy (ART) prescribed by medical professionals,” the agency explained.
NAFDAC emphasised that the product is unregistered and that its ingredients and manufacturing standards are unclear, as the safety, quality, and effectiveness have not been verified through clinical trials.
The regulatory body cautioned that using unverified products like ViroActive+ may lead to treatment failure if patients stop proven HIV medications, cause unknown side effects or toxicity, delay proper medical care, and increase exposure to counterfeit or substandard products.
Product Details:
The product details of the unregistered ViroActive+ are as follows;
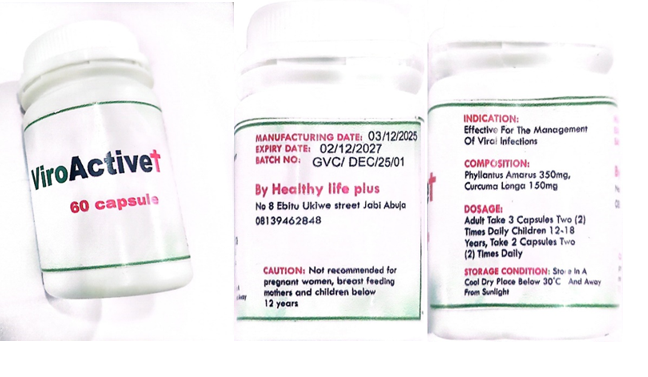
Product name: ViroActive+
Batch Number: GVC/DEC/25/01.
Manufacturing Date: 03/12/2025.
Expiry Date: 02/12/2027.
Product Photo:
NAFDAC ordered its zonal directors and state coordinators to conduct surveillance and remove any unregistered products within their respective zones and states.
The agency urged distributors, retailers, healthcare professionals, and caregivers to exercise caution and vigilance throughout the supply chain to prevent the distribution, sale, and use of the product.
It also advised the public to obtain all medical products from authorised and licensed suppliers and to thoroughly verify the authenticity and physical condition of products before use.
NAFDAC further encouraged healthcare professionals and consumers to report any suspicion of the sale of substandard and falsified medicines or medical devices to its nearest office, by calling 08001623322, or via email at sf.alert@nafdac.gov.ng.
The regulatory body equally advised healthcare professionals and patients to report adverse events or side effects related to the use of medicinal products or devices to its nearest office, through the use of the E-reporting platforms available on its website www.nafdac.gov.ng, via the Med-safety application on Android and IOS, or by email at pharmacovigilance@nafdac.gov.ng.







