NAFDAC Raises Alarm Over Fake Otrivin Nasal Drops in Nigeria

The National Agency for Food and Drug Administration and Control (NAFDAC) has alerted healthcare providers and the public about the circulation of counterfeit and unregistered Otrivin Nasal drops (0.05%) for children and (0.01%) for adults in Nigeria.
NAFDAC affirmed that its Post Marketing Surveillance (PMS) Directorate received a consumer complaint regarding an unregistered Otrivin Nasal Drop being circulated.
According to NAFDAC, investigations and clarification from the Marketing Authorisation Holder (MAH), GlaxoSmithKline, revealed that the company is not currently importing any Otrivin products.
“The MAH also confirmed that all products in circulation are counterfeits,” the agency said, explaining that nasal drops are used to relieve congestion, treat allergies, reduce inflammation, and soothe dryness by delivering medication or saline directly into the nasal passages.
It stressed that the integrity of a product depends on compliance with regulatory requirements, warning that unregistered products lacking regulatory approval pose significant risks to public health, as they may contain harmful, substandard, or ineffective ingredients.
The agency also pointed out that the use of fake products can lead to adverse health outcomes and treatment failures.
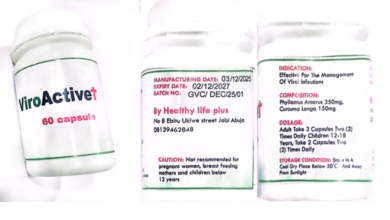
Details of the Counterfeit Products:
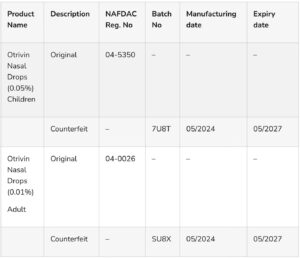
Product Photo:
Original Approved Packaging (Children)
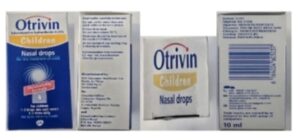
Counterfeit Otrivin (Children)

Original Approved Packaging (Adult)
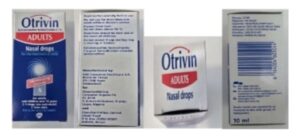
Counterfeit Otrivin (Adult)

NAFDAC directed its zonal directors and state coordinators to conduct surveillance and remove any unregistered or counterfeit products found in their jurisdictions.
The regulatory body urged distributors, retailers, healthcare professionals, and caregivers to exercise caution and vigilance throughout the supply chain to prevent the distribution, sale, and use of substandard products.
It also advised the public to purchase medical products only from authorised and licensed suppliers and to carefully check the authenticity and physical condition of products before use.
NAFDAC encouraged healthcare professionals and consumers to report any suspicion of the sale of substandard and falsified medicines or medical devices to its nearest office, by calling 08001623322, or via email at sf.alert@nafdac.gov.ng.
The agency further urged healthcare professionals and patients to report adverse events or side effects related to the use of medicinal products or devices to its nearest office, through the use of the E-reporting platforms on www.nafdac.gov.ng, via the Med-safety application on Android and IOS, or by e-mail at pharmacovigilance@nafdac.gov.ng.