NAFDAC Raises Alarm Over Fake Herceptin in Nigeria

The National Agency for Food and Drug Administration and Control (NAFDAC) has alerted healthcare professionals and the general public to the circulation of a counterfeit batch of Herceptin 600 mg in Nigeria.
NAFDAC stated that the alert follows official communication from Roche Nigeria confirming a counterfeit case of Herceptin 600 mg with batch number A8519B34 reported in Lagos State.
The agency disclosed that the report was received as a complaint from a customer by the Marketing Authorisation Holder (MAH), Roche Nigeria.
“The counterfeited batch of the product was said to have been offered to a customer by a pharmacist in Lagos at a significantly lower price of NGN 50,000 compared to the standard rate,” NAFDAC said.
The regulatory body explained that, upon investigation, the MAH compared images of the suspected counterfeit Herceptin 600 mg provided by the complainant with the genuine packaging material of the product.
According to NAFDAC, the comparison revealed significant differences from the genuine packaging, including incorrect artwork, a wrong expiry date, and an invalid 2D matrix code.
The agency also noted that lot tracing was not possible, as the batch number A8519B34 is not a genuine Roche batch number, adding that chemical analysis could not be conducted because the physical sample was not available for examination.
NAFDAC explained that Herceptin (trastuzumab) is a prescription medicine used to treat certain types of breast and stomach cancers, stressing that it works by inhibiting cancer cell growth and administered via intravenous infusion or injection, often in combination with chemotherapy.
The agency warned that counterfeit oncology medicines may contain incorrect or no active ingredients, harmful contaminants, or improper dosage strength. Such risks, it said, could lead to treatment failure, disease progression, serious adverse effects, or death.
The details of the counterfeit products are as follows:
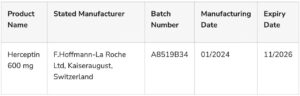
The genuine Herceptin 600 mg is manufactured by F. Hoffmann-La Roche Ltd in Basel, at the Kaiseraugust site in Switzerland.
Product picture
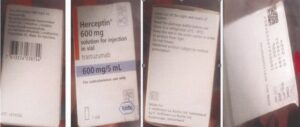
Counterfeit Herceptin 600 mg A8519B34
NAFDAC directed its zonal directors and state coordinators to carry out surveillance and mop up the counterfeit product within their respective zones and states.
The regulatory body advised importers, distributors, retailers, healthcare professionals, and caregivers to exercise caution and vigilance within the supply chain to avoid the importation, distribution, sale, and use of counterfeit products.
It urged the public to obtain all medical products from authorised or licensed suppliers, reiterating that the products’ authenticity and physical condition should be carefully checked.
NAFDAC also advised healthcare professionals and consumers to report any suspected sale of substandard and falsified medicines or medical devices to its nearest office by calling 08001623322 or by email at sf.alert@nafdac.gov.ng.
Healthcare professionals and patients were equally encouraged to report adverse events or side effects related to the use of medicinal products or devices to the nearest NAFDAC office, through the e-reporting platforms available on the NAFDAC website (www.nafdac.gov.ng), via the Med Safety application available for download on Android and iOS stores, or by email at pharmacovigilance@nafdac.gov.ng.







