NAFDAC Raises Alarm on Fake Visitect HIV Test Kits in Nigeria

The National Agency for Food and Drug Administration and Control (NAFDAC) has alerted healthcare providers, patients, and the public to the circulation of counterfeit and parallel-imported, unregistered versions of the VISITECT HIV Advanced Disease Test Kits in Nigeria.
NAFDAC stated that the Marketing Authorisation Holder (MAH) of the product, EURO SPECS International Nigeria Limited, has confirmed the existence of counterfeit and parallel-imported unregistered versions being distributed through unauthorised channels.
“The VISITECT CD4 Advanced Disease assay is a rapid, instrument-free lateral flow test designed to identify patients with severe HIV (CD4 count < 200 cells/µL) within 40 minutes using blood samples. This assay is a vital diagnostic tool for resource-limited settings, helping to triage patients who require immediate advanced care. It offers high sensitivity in detecting low CD4 levels,” the Agency explained.
NAFDAC noted that the integrity of regulated products depends heavily on strict adherence to regulatory requirements, warning that falsified, unlicensed, and unregistered products pose significant risks to public health because they have not been evaluated by the Agency. Consequently, their quality, safety, and efficacy cannot be guaranteed.
The key discrepancies identified between the genuine version (EURO SPECS International Nigeria Limited) and the counterfeit version are as follows:

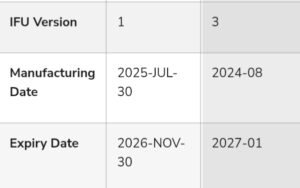
The product label of the counterfeit version displays a three (3) year shelf-life 2024-08 – 2027-01 as against an eighteen (18) months shelf-life approved by NAFDAC.
Product Photo

Fig 1: Approved VISITECT CD4 Advanced Disease Test Kit Photo.

Fig 2 and 3: Counterfeit/Unregistered VISITECT CD4 Advanced Disease Test Kit Photo.
NAFDAC reiterated its commitment to safeguarding public health and assured that it will continue surveillance activities to ensure the quality, safety, and efficacy of medicines, medical devices, and other regulated products circulating in Nigeria.
The Agency ordered its zonal directors and state coordinators to conduct surveillance and remove any counterfeit or unregistered products found within their respective zones and states.
It advised distributors, retailers, healthcare professionals, and caregivers to remain vigilant, carefully verify the authenticity and physical condition of medical products, and ensure that all supplies are sourced only from authorised and licensed distributors.
NAFDAC also urged healthcare professionals to review stocks within hospitals, clinics, and pharmacy stores and to report any suspected falsified or compromised medicines or medical devices to its nearest office, by calling 08001623322, or via email at sf.alert@nafdac.gov.ng.
The regulatory body further encouraged healthcare professionals and patients to report adverse events or side effects related to the use of medicinal products or devices to its nearest office, through the E-reporting platforms available on its website www.nafdac.gov.ng, via the Med Safety application available for download on Android and IOS devices, or by email at pharmacovigilance@nafdac.gov.ng.








3zf3ny