NAFDAC Alerts Public to Meril Diagnostics Medical Devices Flagged by WHO

The National Agency for Food and Drug Administration and Control (NAFDAC) has alerted Nigerians, including healthcare providers and patients, to a Notice of Concern (NOC) issued by the WHO Prequalification Service regarding some NAFDAC-registered medical devices manufactured by Meril Diagnostics Pvt. Ltd., India.
NAFDAC noted that the NOC was issued following significant violations observed during the WHO Prequalification (WHO-PQT) audit of the company’s manufacturing facility.
“This necessitated a declaration that the manufacturer was not operating in line with WHO requirements and applicable quality standards. Hence, the quality and safety of the medical devices are not guaranteed,” the Agency said.
Product Details
The details of the affected medical devices manufactured by Meril Diagnostics Pvt. Ltd in India and marketed by KVATH Int’l Ltd (the Marketing Authorization Holder (MAH)) in Nigeria are as follows:
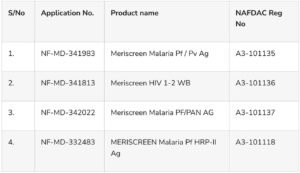
NAFDAC disclosed that while the products have been registered, the MAH (KVATH Int’l Ltd), confirmed that they have not yet been imported into Nigeria due to the company’s current non-operational status following external policy changes.
However, the regulatory body affirmed that if the products are found in the country, they will be considered falsified, counterfeit, and fraudulently imported.
NAFDAC reiterated its committment to safeguarding public health, adding that it will continue surveillance activities to ensure the quality, safety, and efficacy of medicines, medical devices, and other regulated products circulating in Nigeria.
The Agency directed its zonal directors and state coordinators to conduct surveillance and remove any such products found in their respective zones and states.
It advised distributors, retailers, healthcare professionals, and caregivers to exercise caution and vigilance regarding the listed products and to refrain from distributing, selling, or using them.
NAFDAC urged the public to obtain all medical products from authorised or licensed suppliers, stressing that the authenticity and physical condition of such products should be carefully checked before use.
The regulatory body also encouraged healthcare professionals to review stocks within hospitals, clinics, and pharmacy stores and report any suspected falsified or compromised products (medicines or medical devices) to the nearest NAFDAC office, call 08001623322, or send an email to sf.alert@nafdac.gov.ng.
Health professionals and patients are equally advised to report adverse events or side effects related to the use of medicinal products or devices to the nearest NAFDAC office, through the e-reporting platforms available on the NAFDAC website www.nafdac.gov.ng, via the Med Safety application available for download on Android and IOS stores, or by email at pharmacovigilance@nafdac.gov.ng.







