NAFDAC Raises Alarm Over Fake Avastin in Nigeria

The National Agency for Food and Drug Administration and Control (NAFDAC) has alerted healthcare providers and the public to the circulation of counterfeit Avastin 400mg in Nigeria.
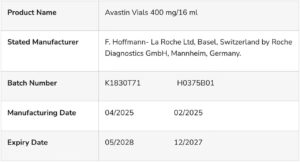
According to NAFDAC, the Marketing Authorisation Holder (MAH), Roche, reported that an oncologist from a local hospital expressed concern about potentially counterfeit Avastin vials (400 mg/16 ml), batch numbers K1830T71 and H0375B01.
The agency explained that the complaint sample was compared with a genuine retained sample in the EFA (English, French, and Arabic) make-up presentation, because the GTIN (Global Trade Item Number) of the complaint sample’s folding box corresponded with the EFA make-up presentation.
Differences Observed During Investigation
– The batch number does not exist in the Roche database, making lot tracing impossible.
– The artwork contained incorrect text and spelling errors.
– Variable data was incorrect, and the placement was not consistent.
– The Tamper Evidence labels do not correspond with genuine Roche material.
– The complaint sample serial number does not correspond with any genuine Roche serial number.
While affirming that the findings indicate the presence of counterfeit packaging, NAFDAC stated that Avastin (bevacizumab) is indicated for the treatment of recurrent glioblastoma in adults.
The regulatory body noted that the drug is a tumour-starving (anti-angiogenic) therapy, warning that the illegal marketing of medicines and counterfeit products poses a serious risk to public health, as such products do not guarantee safety, quality, or efficacy due to non-compliance with regulatory provisions.
Product details
The details of the counterfeit products are as follows:
NAFDAC directed its zonal directors and state coordinators to carry out surveillance and mop up the counterfeit product within their respective zones and states.
The agency also advised importers, distributors, retailers, healthcare professionals, and caregivers to exercise caution and vigilance within the supply chain to prevent the importation, distribution, sale, and use of counterfeit products.
It urged the public to obtain medical products only from authorised and licensed suppliers, adding that the authenticity and physical condition of products should be carefully checked before purchase or use.
The regulatory body further advised healthcare professionals and consumers to report any suspicion of the sale of substandard and falsified medicines or medical devices to its nearest office, by calling 08001623322, or by email at sf.alert@nafdac.gov.ng.
Healthcare professionals and patients are equally encouraged to report adverse events or side effects related to the use of medicinal products or devices to the nearest NAFDAC office, through the e-reporting platforms available on www.nafdac.gov.ng, via the Med-Safety mobile application available on Android and IOS stores, or by email at pharmacovigilance@nafdac.gov.ng







